SECuRE trial update: Another participant with undetectable PSA & negative PSMA PET
PR Newswire
SYDNEY, Feb. 23, 2026
HIGHLIGHTS
- Another participant with metastatic castration-resistant prostate cancer (mCRPC) in the Cohort Expansion Phase (Phase II; 8 GBq of 67Cu-SAR-bisPSMA per cycle) of the SECuRE trial (NCT04868604)1 achieved undetectable disease as assessed by prostate-specific antigen (PSA) and prostate-specific membrane antigen (PSMA) positron emission tomography (PET).
- Undetectable disease by PSA was achieved after the first cycle, followed by a negative PSMA PET after the second cycle of 67Cu-SAR-bisPSMA.
- Participant was initially diagnosed with prostate cancer 15 years ago, had metastatic disease since 2020 and experienced progressive mCRPC in 2025.
- All related adverse events (AEs) were mild (Grade 1), most of which resolved after the treatment. No haematological or renal AEs were observed to date.
- This is the fifth patient to achieve undetectable disease by radiographic assessment following 67Cu-SAR-bisPSMA treatment.
- The previous participant in the same Cohort Expansion Phase who achieved undetectable PSA after three cycles of 8 GBq 67Cu-SAR-bisPSMA2 (announced on the 15 January 2026) continues to demonstrate undetectable disease on PSMA PET after the fourth cycle.
- The SECuRE trial will continue enrolment into the Cohort Expansion Phase with planned completion of recruitment in 2026. Phase III registrational trial planning is ongoing based on data generated to date.
SYDNEY, Feb. 23, 2026 /PRNewswire/ -- Clarity Pharmaceuticals (ASX: CU6) ("Clarity" or "Company"), a clinical-stage radiopharmaceutical company with a mission to develop next-generation products that improve treatment outcomes for patients with cancer, is pleased to announce that yet another participant with mCRPC in the Cohort Expansion Phase (Phase II) of the SECuRE trial achieved undetectable PSA and negative PSMA PET. The undetectable PSA was measured following the first cycle and the negative PSMA PET was reported following the second cycle of 67Cu-SAR-bisPSMA (8 GBq each cycle).
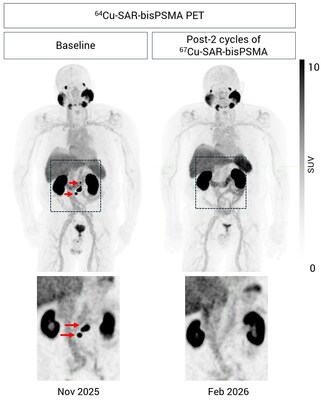
The participant is a 76-year-old man who was initially diagnosed with prostate cancer 15 years ago. He had radical prostatectomy to treat the primary disease and radiotherapy for local recurrence, having progressed to metastatic disease in 2020. Previous systemic anti-cancer treatments included an androgen receptor pathway inhibitor (ARPI) and androgen deprivation therapy (ADT). In 2025, the disease progressed further, and he was enrolled into the Cohort Expansion Phase of the SECuRE study with a baseline PSA of 3.25 ng/mL. Seven weeks after his first cycle of 67Cu-SAR-bisPSMA, this participant achieved undetectable PSA levels. He proceeded to receive one more cycle of 67Cu-SAR-bisPSMA, and no disease was observed on PSMA PET following the second dose (Figure 1). This participant exhibited mild (Grade 1) related AEs, including altered taste, dry eyes, eye pain, fatigue and salivary gland soreness (all resolved except fatigue). No haematological or renal AEs were observed to date.
This new case represents the fifth participant to achieve undetectable disease by radiographic assessment following 67Cu-SAR-bisPSMA treatment in Clarity's SAR-bisPSMA theranostic program (3 participants who received up to four cycles of 8 GBq, and 2 participants who received up to three cycles of 12 GBq)2,3,4.
Update on previously reported participant with no detectable disease following 67Cu-SAR-bisPSMA treatment
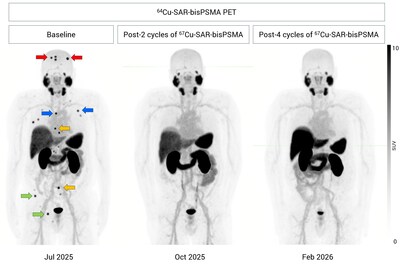
Follow-up on the previously reported participant from the Cohort Expansion Phase (no detectable disease and absence of prior bone metastasis following three cycles of 8 GBq 67Cu-SAR-bisPSMA treatment)2, demonstrated that his PSA remains undetectable, with no disease identified on his latest PET conducted 1 month after the administration of the fourth cycle (Feb 2026; Figure 2). Following the first three cycles of 67Cu-SAR-bisPSMA, the participant exhibited mild (Grade 1) related AEs, most of which were gastrointestinal events, with no haematological or renal AEs2. Notably, no new safety signals have been observed during and since the administration of the fourth cycle to date.
The interim data from this Phase II continues to confirm the favourable safety profile and promising efficacy seen in previous cohorts of SECuRE3 and supports the continuation of the trial with the aim to progress to a registrational Phase III study.
Clarity's Executive Chairperson, Dr Alan Taylor, commented, "The momentum of data we are generating with our lead SAR-bisPSMA product in both theranostic and diagnostic trials is strong, with excellent results to date on all fronts. We are beyond excited to see yet another patient achieve undetectable disease following their 67Cu-SAR-bisPSMA treatments. What is particularly outstanding is that this new patient presented with undetectable PSA just 7 weeks after his first 8 GBq cycle, with a negative PSMA PET being reported after the second cycle. This result is particularly impressive given this participant has been battling prostate cancer for 15 years and is now free of any detectable disease based on PSA and PET assessments with only mild (Grade 1) AEs.
"While this outcome is extraordinary, we now know this is not just luck or coincidence. Despite the number of participants in the SECuRE trial being relatively small, this is now the fifth time we have seen a patient present with undetectable disease following their 67Cu-SAR-bisPSMA treatments2,3,4, and study recruitment is still ongoing. We are also excited to see that the previous participant in the Cohort Expansion Phase of the SECuRE trial to achieve undetectable disease observed by anatomical and molecular imaging2 continues to show undetectable disease during his last follow up in February 2026, following the fourth 67Cu-SAR-bisPSMA cycle. This is especially encouraging as this participant had bone metastasis at study entry and now continues to report excellent quality of life after his treatment with 67Cu-SAR-bisPSMA."
"Our methodical commitment to science and the highest standard of clinical development underpins our achievements to date. Combining the optimised bivalent "bis" structure with the benefits offered by the copper isotope pairing, enabled by the proprietary sarcophagine (SAR) chelating technology, is key to the benefits we are seeing from the SAR-bisPSMA products. The most recent data released from the Co-PSMA study clearly establishes that 64Cu-SAR-bisPSMA considerably outperforms its competitors in detecting prostate cancer recurrence and sheds light on the importance of the improved lesion detection, where the diagnostic benefits translate into enhanced patient management5. Our registrational diagnostic AMPLIFY6 and CLARIFY7 trials with 64Cu-SAR-bisPSMA are also nearing the end of recruitment, getting us closer to commercialisation. We now look forward to progressing the SECuRE trial recruitment and have already initiated the planning for a registrational Phase III trial, as we hope to see more people benefit from our unique theranostic product. Having three Fast Track Designations (FTDs) for SAR-bisPSMA gives us confidence to persevere harder than ever, and positive interactions with the US Food and Drug Administration (FDA) confirm that we are on the right track. Our team is motivated and driven to progress SAR-bisPSMA to the market through the entirety of the prostate cancer journey, from first detection to late-stage metastatic disease. As we continue to generate exceptional data, we believe SAR-bisPSMA is well positioned to fully exploit the current treatment and diagnostic challenges in radiopharmaceuticals and improve treatment outcomes for patients with cancer."
About the SECuRE trial
The SECuRE trial (NCT04868604)1 is a Phase I/IIa theranostic trial for identification and treatment of participants with PSMA-expressing mCRPC using 64Cu/67Cu-SAR-bisPSMA. 64Cu-SAR-bisPSMA is used to visualise PSMA-expressing lesions and select candidates for subsequent 67Cu-SAR-bisPSMA therapy. The trial is a multi-centre, single arm study, planning to enroll approximately 54 participants in the US. The overall aim of the trial is to determine the safety and efficacy of 67Cu-SAR-bisPSMA for the treatment of prostate cancer.
The SECuRE trial consists of the Dose Escalation (Phase I) and Cohort Expansion (Phase II) Phases. Based on the data from the Dose Escalation Phase, which demonstrated a favourable safety profile and efficacy of 67Cu-SAR-bisPSMA, the SECuRE trial progressed to the Cohort Expansion at an 8 GBq dose level as per the Safety Review Committee (SRC) recommendation (up to 6 cycles per patient in total)3.
Cohort 2 of the Dose Escalation phase of the trial, where participants were dosed with 8 GBq of 67Cu-SAR-bisPSMA, demonstrated a very low rate of related AEs while all three participants achieved PSA declines of 80% or more (PSA80)3. The Dose Escalation Phase also showed high PSA response rates of the mCRPC in the pre-chemotherapy setting with a favourable safety profile: 92% of pre-chemotherapy participants (12/13) demonstrated PSA drops greater than 35%, 61.5% (8/13) of participants achieved PSA reductions greater than 50%, and 46.2% (6/13) of participants achieved PSA reductions of 80% or more3. These results supported the progress of the trial to its Cohort Expansion Phase using 8 GBq multi-dose in participants who had not received chemotherapy in the mCRPC setting.
Recruitment is currently ongoing into the Cohort Expansion Phase which will include 24 participants. A subset of participants will be treated with the combination of 8 GBq of 67Cu-SAR-bisPSMA with enzalutamide (ARPI), in line with the positive results from the Enza-p trial8 and previous discussions with and advice from key global medical experts in the field of prostate cancer.
About SAR-bisPSMA
SAR-bisPSMA derives its name from the word "bis", which reflects a novel approach of connecting two PSMA-targeting agents to Clarity's proprietary SAR technology that securely holds copper isotopes inside a cage-like structure, called a chelator. Unlike other commercially available chelators, the SAR technology prevents copper leakage into the body. SAR-bisPSMA is a Targeted Copper Theranostic that can be used with isotopes of copper-64 (Cu-64 or 64Cu) for imaging and copper-67 (Cu-67 or 67Cu) for therapy.
67Cu-SAR-bisPSMA and 64Cu-SAR-bisPSMA are unregistered products. The safety and efficacy of 67Cu-SAR-bisPSMA and 64Cu-SAR-bisPSMA have not been assessed by health authorities such as the US FDA or the Therapeutic Goods Administration (TGA). There is no guarantee that these products will become commercially available.
About Prostate Cancer
Prostate cancer is the second most common cancer diagnosed in men globally and the fifth leading cause of cancer death in men worldwide9. Prostate cancer is the second-leading cause of cancer death in American men. The American Cancer Institute estimates there will be about 333,830 new cases of prostate cancer in the US in 2026 and around 36,320 deaths from the disease10.
About Clarity Pharmaceuticals
Clarity is a clinical-stage radiopharmaceutical company focused on the treatment of serious diseases. The Company is a leader in innovative radiopharmaceuticals, developing TCTs based on its SAR Technology Platform for the treatment of cancers.
www.claritypharmaceuticals.com
For more information, please contact:
Clarity Pharmaceuticals
Dr Alan Taylor Lisa Sadetskaya
Executive Chairperson Director, Corporate Communications
ataylor@claritypharm.com lisa@claritypharm.com
References |
1. ClinicalTrials.gov Identifier: NCT04868604, https://clinicaltrials.gov/ct2/show/NCT04868604 |
2. Clarity Pharmaceuticals. SECuRE trial to continue with no modifications to protocol following Safety Review Committee meeting. https://www.claritypharmaceuticals.com/news/secure-src/ |
3. Clarity Pharmaceuticals. SECuRE trial update: 92% of pre-chemo participants experience greater than 35% drop in PSA levels across all cohorts. Cohort Expansion Phase commences. https://www.claritypharmaceuticals.com/news/secure-update/ |
4. Clarity Pharmaceuticals. Copper-67 SAR-bisPSMA updates. https://www.claritypharmaceuticals.com/news/secure-update/ |
5. Clarity Pharmaceuticals. Co-PSMA: Cu-64 SAR-bisPSMA more than doubled prostate cancer lesion and patient detection vs. Ga-68 PSMA-11 in head-to-head trial. https://www.claritypharmaceuticals.com/news/abstract-eau-2026/ |
6. ClinicalTrials.gov Identifier: NCT06970847. https://clinicaltrials.gov/study/NCT06970847 |
7. ClinicalTrials.gov Identifier: NCT06056830. https://clinicaltrials.gov/study/ NCT06056830 |
8. Emmett L et al. ENZA-p Trial Investigators; Australian and New Zealand Urogenital and Prostate Cancer Trials Group. Overall survival and quality of life with [177Lu]Lu-PSMA-617 plus enzalutamide versus enzalutamide alone in metastatic castration-resistant prostate cancer (ENZA-p): secondary outcomes from a multicentre, open-label, randomised, phase 2 trial. Lancet Oncol. 2025 Mar;26(3):291-299. doi: 10.1016/S1470-2045(25)00009-9. |
9. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. https://acsjournals.onlinelibrary.wiley.com/doi/10.3322/caac.21660 |
10. American Cancer Society: Key Statistics for Prostate Cancer. https://www.cancer.org/cancer/prostate-cancer/about/key-statistics.html |
This announcement has been authorised for release by the Executive Chairperson.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/secure-trial-update-another-participant-with-undetectable-psa--negative-psma-pet-302694256.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/secure-trial-update-another-participant-with-undetectable-psa--negative-psma-pet-302694256.html
SOURCE Clarity Pharmaceuticals